The Southwest National Primate Research Center (SNPRC) serves as a national resource for investigators who are performing nonhuman primate research. There are three main services provided by the SNPRC: animal sales, biomaterials requests, and research requests. SNPRC breeds Indian origin rhesus macaques, olive baboons, and common marmosets. Investigators interested in performing research studies with these animals, obtaining tissues from these animals, or acquiring these animals from our center should fill out a request form below.
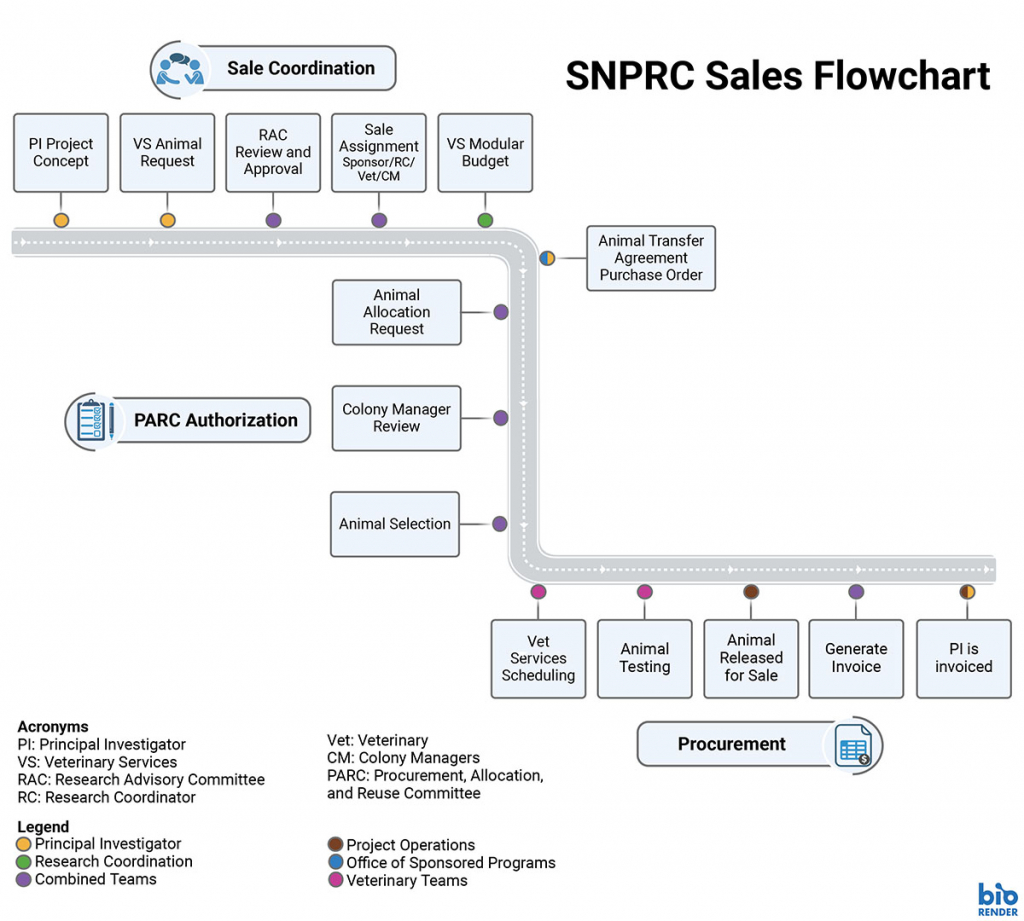
Animal Sales
Researchers can acquire macaques, baboons, and marmosets from our colonies. Please note that requests are subject to review. Please note that availability of geriatric animals, marmosets, and macaques (for non HIV-related studies) is limited
Downloadable PDF:
Make an Animal Purchase Request
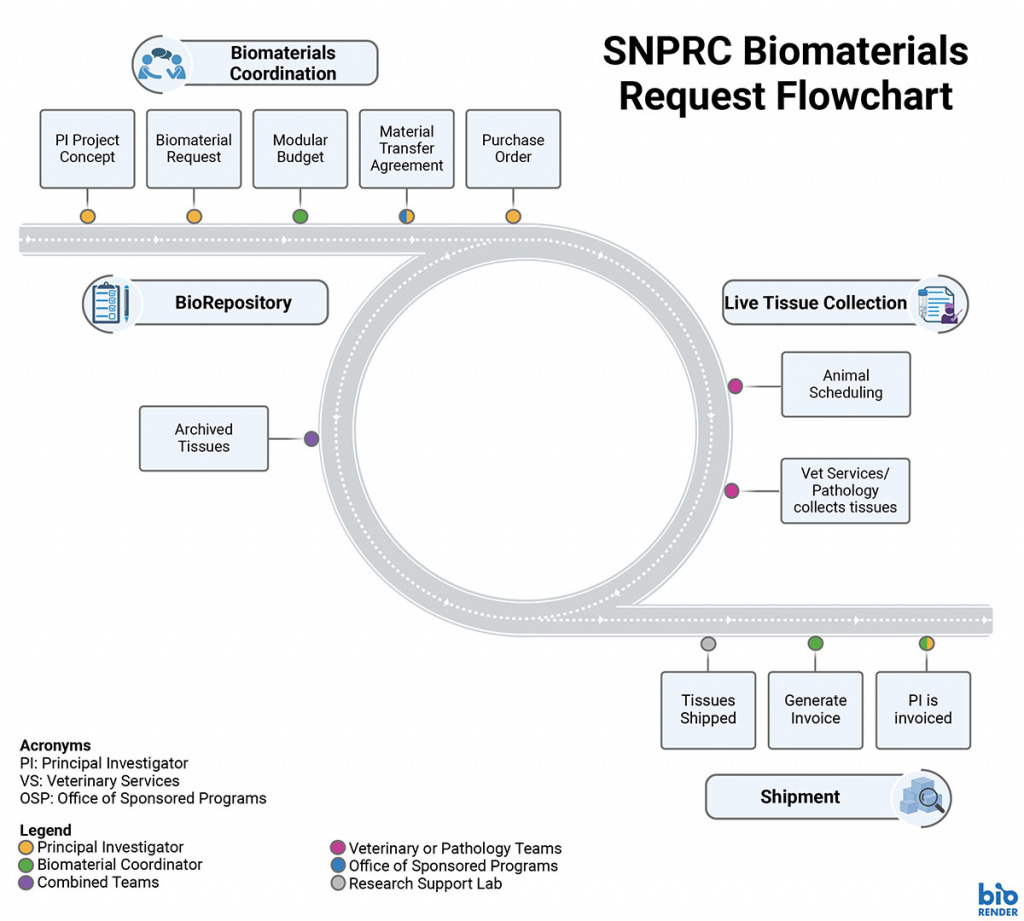
Biomaterials Requests
SNPRC maintains a biorepository with frozen tissue samples from mostly healthy nonhuman primates collected at necropsy. In addition fresh tissues can be requested from animals taken at the time of necropsy. Many fresh noninvasive tissues can also be collected from colony animals.
Downloadable PDF:
Make a Biological Materials Request
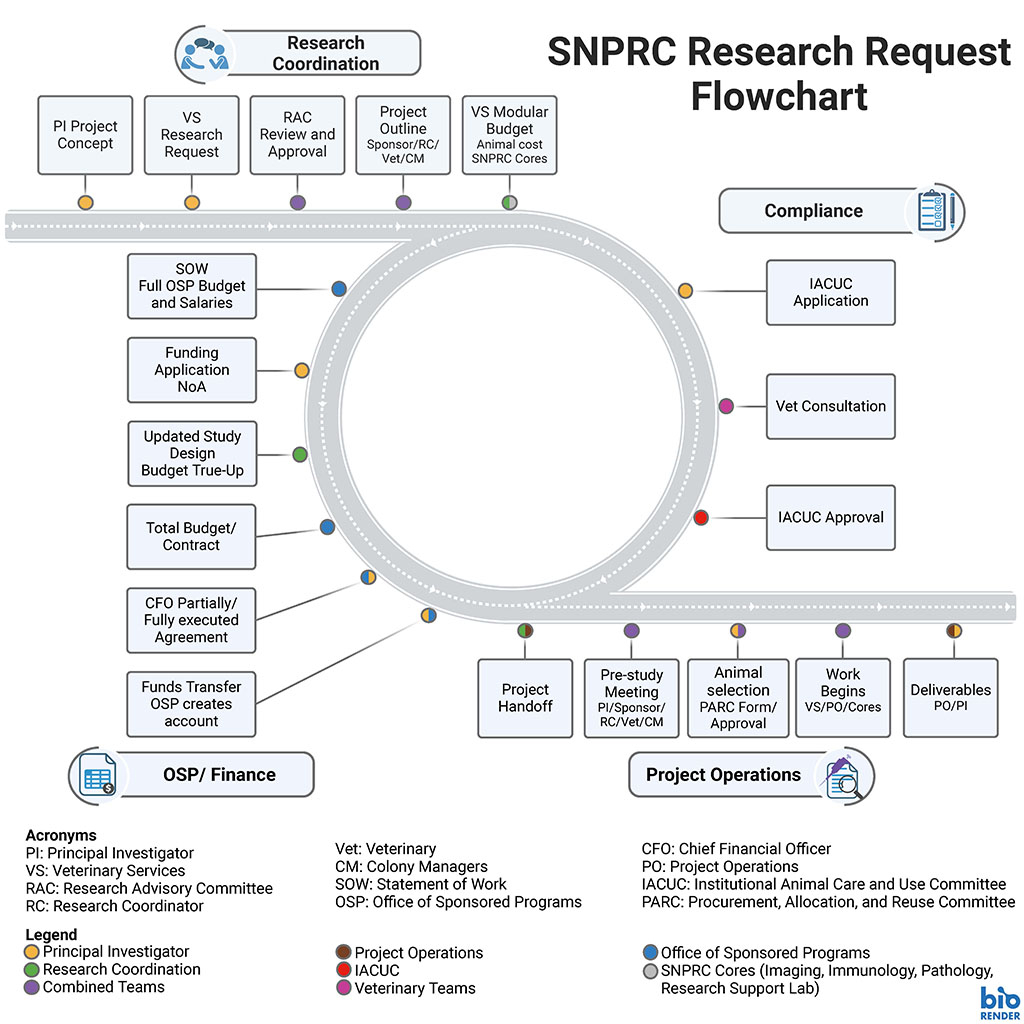
Research Requests
Investigators can request to do research projects at SNPRC. SNPRC has internal SNPRC Sponsors, Research Coordinators, Scientific Cores, and a Support Laboratory. These groups assist with every step of the research process including study design, budgeting, grant submission, contracts, test article preparation, sample processing, imaging, immunology, pathology, molecular assays, and reporting.
Downloadable PDF:
Still have questions?
Please contact the Assistant Director of Research, Angélica M. Olmo-Fontánez, Ph.D. .